top of page
Browse by category
Search


FitForMe launches new supplements addressing protein deficiency, digestive issues and nutritional deficiencies during obesity treatment
More than one billion people worldwide are living with obesity. Lifestyle intervention programmes, GLP-1 medications and bariatric surgery can be transformative, but they do not exist in a nutritional vacuum. Deficiencies are common among people affected by obesity before treatment begins, and surgery or medication can make them harder to ignore. Patients arrive underprepared, find little nutritional support built into their care, and are largely left to manage the consequenc


Olympus gains FDA clearance for POWERSEAL Open Extended Jaw Device
Olympus Corporation has received FDA 510(k) clearance for the POWERSEAL Open Extended Jaw Sealer/Divider. This latest edition to the Olympus POWERSEAL advanced bipolar surgical energy portfolio supports clinical needs for precision, efficiency and control during surgery. POWERSEAL Open Extended Jaw Sealer/Divider The POWERSEAL Open Extended Jaw Sealer/Divider joins three 5mm devices in the POWERSEAL portfolio. These 5mm devices are offered in three jaw configurations, each av


J&J gains CE Mark Approval for ETHICON 4000 Stapler
Johnson & Johnson has received European CE Mark approval for its new ETHICON 4000 Stapler, an advanced surgical stapler designed to deliver reliable staple line integrity across a wide range of tissue thickness. ETHICON 4000 Stapler With proprietary 3D Stapling Technology, a redesigned end-effector and streamlined reload options, the ETHICON 4000 and ETHICON 3D Reloads are certified for use in open and laparoscopic surgery across the European Union. The new advanced stapling


Reach Surgical gains CE Mark for iREACH IRIS, the first powered reusable stapler with Real-Time Firing Curve and 90° Articulation
Reach Surgical, the surgical solutions division of Genesis MedTech, has received CE Mark approval for iREACH IRIS, a next-generation powered stapling platform designed to deliver Real-Time Firing Curve and up to 90° articulation during surgery. iREACH IRIS Reusable Stapler with Real-Time Firing Curve™ and 90° Articulation Unlike conventional powered staplers that provide general firing feedback, iREACH IRIS Real-Time Firing Curve provides a continuous, location-specific force


FDA approves Lilly's Foundayo (orforglipron) GLP-1 pill for weight loss
The FDA has approved Eli Lilly and Company’s Foundayo (orforglipron) for adults with obesity or overweight with weight-related medical problems. Foundayo is a once-daily small molecule (non-peptide) oral glucagon-like peptide-1 receptor agonist (GLP-1ra) that can be taken any time of the day without restrictions on food and water intake. "People living with obesity need treatment options that meet them where they are – and for many, a once-daily pill that can be taken with n

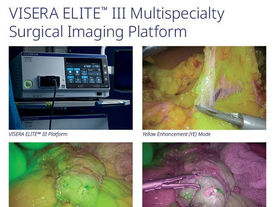
Olympus launches next-generation VISERA ELITE III surgical imaging platform in US
Olympus Corporation has launched the VISERA ELITE III surgical imaging platform designed to enhance surgical visualization, workflow efficiency and multispecialty flexibility, in the US. The VISERA ELITE III platform is a significant leap forward in surgical imaging and the third major co-developed product by SONY Olympus Medical (SOMED), a joint venture established in 2013. The VISERA ELITE III surgical imaging platform offers advanced features including Yellow Enhancement (


FDA approves Allurion Gastric Balloon System
The FDA has approved the premarket approval (PMA) application for the Allurion Gastric Balloon System (AGBS), featuring the Allurion Smart Capsule. The Allurion Program is a weight-loss platform that combines the FDA PMA approved Allurion Gastric Balloon System, featuring the Allurion Smart Capsule, with the Allurion Virtual Care Suite, including the Allurion Mobile App for consumers featuring the Iris AI platform, Allurion Insights for healthcare providers, and the Allurion


Europe approves 7.2 mg injectable Wegovy for adults with obesity
The European Commission has granted final approval of a higher 7.2 mg maintenance dose of Wegovy (semaglutide) for adults who may need extra help losing weight. It is now approved in all 27 countries in the European Union. This gives doctors another option to help adults who need more weight loss after being on the 2.4 mg dose. The approval is based on a positive opinion from the European Medicines Agency’s scientific committee (CHMP) on 12 December 2025. The approval means t


FDA approves Novo Nordisk's oral GLP-1 for weight loss
The FDA has approved Novo Nordisk’s once-daily Wegovy pill, the first oral GLP-1 medicine for obesity in the US. The Wegovy pill can be used with a reduced calorie diet and increased physical activity for adults with obesity, or with overweight who also have weight-related medical problems, to help them lose weight and keep it off. The pill is also indicated to reduce the risk of major adverse cardiovascular events (MACE) such as death, heart attack, or stroke in adults with


Novo Nordisk files for FDA approval for CagriSema, the first once-weekly combination of GLP‑1 and amylin analogues
Novo Nordisk has submitted a New Drug Application (NDA) to the FDA for once-weekly CagriSema (cagrilintide 2.4 mg and semaglutide 2.4 mg) injection, to be used with a reduced-calorie diet and increased physical activity, to reduce excess body weight and maintain weight reduction long term in adults with obesity or overweight in the presence of at least one weight-related comorbid condition. CagriSema is a fixed-dose combination of a long-acting amylin analogue, cagrilintide 2
Browse by tag






bottom of page
