top of page
Browse by category
Search


Gender, metabolism and genetics may play a crucial role in lower cancer risk after bariatric surgery
Two studies, led by researchers from the University of Gothenburg, have provided clues to why substantial and sustained weight-loss has been linked to a reduced risk of cancer and cancer-related death, mainly in women. “Our results suggest that there are biological differences between individuals that affect how much cancer risk is reduced after weight loss,” said Dr Kajsa Sjöholm, associate professor at the University of Gothenburg. “By understanding these differences, we wi


Triple agonist retatrutide 12mg results in 30.3% weight loss at 104 weeks
Results from TRIUMPH-1, a Phase 3 clinical trial evaluating the efficacy and safety of retatrutide in adults with obesity or overweight and at least one weight-related comorbidity and without diabetes, have revealed that people with severe obesity on the highest dose (12mg) lost on average 30% of their body weight over two years. In addition, at 80 weeks, all doses of retatrutide (4mg, 9mg and 12mg) met the primary and key secondary endpoints for obesity, delivering clinicall


BARI-STEP: Semaglutide helps patients who do not respond to bariatric surgery
Outcomes from the BARI-STEP trial, led by Dr Janine Makaronidis at the University College London Hospitals (UCLH) Centre for Obesity Research, has concluded that the weight loss drug semaglutide helps patients who do not respond to bariatric surgery. Janine Makaronidis “These findings strengthen the case for using semaglutide in people who do not respond well to bariatric surgery, offering an alternative to repeat operations,” said Makaronidis. “They also suggest that combini
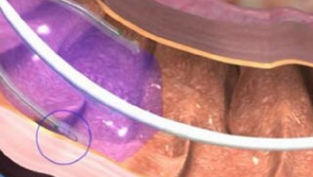
Duodenal mucosal resurfacing may prevent weight recurrence following GLP-1 discontinuation
An outpatient procedure may offer a way for the estimated 70% of people who discontinue popular weight-loss drugs to avoid regaining weight, according to a study, ‘Duodenal mucosal resurfacing prevents weight regain after tirzepatide withdrawal: REMAIN-1 multicenter, randomized, double-blind, sham-controlled clinical trial—midpoint cohort results’, presented at Digestive Disease Week 2026. Nearly one in five adults with obesity have taken a glucagon-like peptide-1 (GLP-1), an


ForePass replicates effects of bariatric surgery and outperforms GLP-1
The ForePass endoscopic metabolic bypass platform reproduced insulin sensitivity levels observed following biliopancreatic diversion (BPD) while substantially outperforming semaglutide (Ozempic/Wegovy) in weight control in a randomised preclinical study, according to a study led by Professor Ivo Boskoski, Professor of Digestive Endoscopy at Università Cattolica del Sacro Cuore Facoltà di Medicina e Chirurgia, Rome, Lazio, Italy. ForePass endoscopic metabolic bypass platform


Metabolic surgery may reverse MASH cirrhosis
Researchers at Cleveland Clinic have demonstrated that metabolic dysfunction-associated steatohepatitis (MASH) cirrhosis regression can occur following metabolic and bariatric surgery (MBS). The study, “Can Metabolic Surgery Regress MASH Cirrhosis? Evidence From Paired Biopsies,” was presented at the 2026 Digestive Disease Week Annual Meeting. In a retrospective cohort of patients with biopsy-proven compensated MASH cirrhosis undergoing MBS, investigators evaluated long-term
Browse by tag






bottom of page
